Yes, this is a Q&A but I need to "vent" all the random facts I don't know on some platform, b/c I've about had it. I'll only be "editing" this post, so this thread will go into obscurity for everyone but me. Thanks for understanding.
1. Endocytosis requires ATP. Seriously? Did not realize that endocytosis requires a lot of ATP in order to rearrange the cytoskeleton of a cell and form vesicles.
2. Kaplan: Sigmoidal Growth Curves of Microorganisms
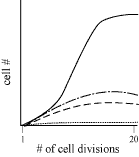
From where? 'Cause it ain't in the book.
3. Not a specific concept, but a task I seem to miss: If I can't find a way to answer with outside knowledge, look in the passage for a situation to connect a novel equation (likely in the passage) with the information given in the passage. B/c finding the relevant info in the passage is the "hard part" explicitly write down the information you think may need.
4.) Entropy (& Enthalpy) is an extensive property. That means I need to multiply the entropy values of each compound by its coefficient. Pressure is an intensive property:
theseeker:" Imagine a vessel containing n moles of gas. Measure the entire vessel, and pressure is P. What is the pressure of the top third of the vessel? How about the bottom tenth? How about the three molecules of gas in the exact center of the vessel? The pressure in the vessel is always P, independently of how much of the gas you are measuring."
5.) Linear mass density = mass/ length , can go up by increasing density, mass, or thickness of strings
6.) Like this point Princeton made about ideal gas intersecting at -273.5 Celcius and 0 Kelvins on P vs. T (or same could be said for V vs. T) graphs. The smaller slope line represents the compound with a greater relative molar mass. PV =nRT can be used to determine slope.

7. AAMC6 had question about terminology transitional vs. rotational vs. just "equilibrium" that made me understand it better.
Transitional equilibrium= sum of external forces is zero. Rotational equilibrium = sum of external torque is zero. And equilibrium is when a body is BOTH in transitional and rotational equilibrium.
8. Oh yeah, platelets (& mature RBCs of course) don't have nuclei and the origin of the coelom is the mesoderm (know it!) & The appendix is in the large intestine
9. Yes, like dissolves like except for a major exception: Acids dissolve bases and Bases dissolve acids (thank LeChatelier). Don't get it twisted.
So which of the following substances is most likely to be more soluble in 1.0 M HCl than in 1.0M NaOH: HI or Pb(OH)2? Pb(OH)2.
10.) Insulation for an electrical current is ALWAYS needed. If an answer choice suggest that: to make less insulation necessary or to minimize insulation, it's useless answer choice.
11.) Just like the catalyst increase the rate of a reaction, an increase in current and/or voltage in an electrochemical cell will increase rate at which a metal at the cathode is produced (not the amount--only amount of starting material can influence amount).
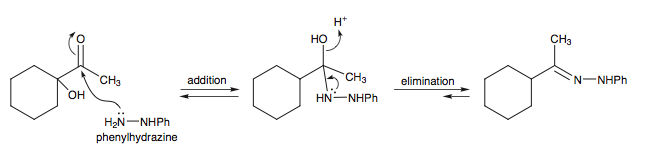
12.) High-yield reaction Converting a ketone ---> imine using hyrdrazone participating in a 2-step addition, elimination rx
13.) Epigenetic development of a gene: modulates gene expression such that one gene can lead to expression of several proteins--so more variation at the RNA level.
14.) Watch out for classic tell-tale sign of a recessive disease: 2 unaffected parents produce an affected child.
15.) Need to remember milliEquivalents equation = mg x valence / (formula weight).
What does an equivalent value tell? It gives you the number of univalent counter ions needed to react with each molecule of substance. So HCl has 1 equivalent of a mole meaning that 1 mole H+ ion is needed to react with 1 mole of a Cl- ion.
14.) telltale of a Sn1 or E1 reaction: rx takes place in acidic conditions. Carbocation can only exists in acidic solutions.
15.) nitric oxide can is capable of diffusing across the plasma membrane & no ions can diffuse across membranes-b/c of the polarity of ions, they need channels
16.) CCK is released by small intestine (by duodenum and jejenum) and acts on the gall bladder to stimulate release of bile.
17.) Double displacement = metathesis (when all ions switch partners kind of like the Shania Twain situation)
18.) Intermolecular forces, strongest to weakest: hydrogen bonding, dipole-dipole, london dispersion forces
But beyond that, hydrogen bonding is well understand. But what about dipole-dipole forces? This occurs when 2 polar bonds from two different molecules attract one another. I got a question wrong that helped m understand this: Dipoles arise from an electronegative bond involved in a polar bond (such as oxygen or nitrogen) pulls electron density from another atom also involved in a polar bond. London dispersion forces are fleeting, make-shift interactions that only arise from electron density temporarily shifting to one side of the molecule.
19.) Increasing the temp will ALWAYS increase rate of rx. So increase in temp = increase in rate of formation of product.
20.) Fats are absorbed into lacteals in the small intestine. Unlike carbohydrate and amino acids, they bypass the hepatic portal system (the liver circulatory system) and head directly into the system circulation via the thoracic duct. And once they're in the bloodstream, fats are packaged into lipoproteins.
21.) Important to look at solvents when determining nature of reaction. For example, Dimethyl sulfoxide (DMSO) is a polar aprotic solvent--boom w/ primary alkyl halide it's an SN2- concerted mechanism w/ neutral intermediate and bimolecular rx.
22.) Pretty much every major carbohydrate is an aldose, except fructose. Fructose is a ketose, whereas glucose, galactose and mannose are all aldoses. The same sugars in different optical families are enantiomers (e.g. D-glucose & L-glucose), all the other same-carbon sugars are disatereomers.
23.) An IR spectrum results when chemical bonds b/w two atoms absorb infrared radiation as a result of interatomic bind vibrations. To give a line in an IR spectrum, remember that the molecule must exhibit a change in dipole moment during vibration. (So Cl2, for example, would not produce an IR spectrum).
24.) This is a momentum problem, I took some time to do with math--but the math is not necessary! So I'll review the problem here.
"An 80 kg ice skater, standing on an essentially frictionless frozen pond, propels himself forward by throwing a 5 kg mass in the opposite direction with a speed of 1m/s. The skater is then propelled forward at a speed of 1/16 m/s. The total momentum o the skater and mass when both are moving is: 0 kg m/s.
The principle of conservation of momentum states that when the net impulse of the external forces acting on a system is zero, the total momentum of the system remains constant. So, I could have considered the skater the mass a system. Before the skater throws the mass, neither has a velocity and so the momentum is zero. The skater was standing and holding the stationary mass as well.
25.) Not new, but I keep forgetting. Primary spermatocytes are diploid. Primarty spermatocytes undergo meiosis I to form haploid secondary spermatocytes.
26.) AAMC listed these frequencies that are important: C≡N = 2250 , C-O: 1000-1300 , C=O : 1700-1850, O-H: 3200-3500
27.) Amides are formed from amines via a nucleophilic acyl substitution.
28.) Important to keep in mind that antibodies are proteins and that T-cells, unlike B-cells, do not recognize free floating antigens
29.) The most effective way to demonstrate necessity is to test for the occurrence of a specific outcome in the absence of the agent whose necessity is being tested.
30.) Macrophages can activate T-helper cells when it presents is antigens to T-cells, it also produces cytokines.
1. Endocytosis requires ATP. Seriously? Did not realize that endocytosis requires a lot of ATP in order to rearrange the cytoskeleton of a cell and form vesicles.
2. Kaplan: Sigmoidal Growth Curves of Microorganisms
You should recall that the growth curves of microorganisms are sigmoidal.

From where? 'Cause it ain't in the book.
3. Not a specific concept, but a task I seem to miss: If I can't find a way to answer with outside knowledge, look in the passage for a situation to connect a novel equation (likely in the passage) with the information given in the passage. B/c finding the relevant info in the passage is the "hard part" explicitly write down the information you think may need.
4.) Entropy (& Enthalpy) is an extensive property. That means I need to multiply the entropy values of each compound by its coefficient. Pressure is an intensive property:
theseeker:" Imagine a vessel containing n moles of gas. Measure the entire vessel, and pressure is P. What is the pressure of the top third of the vessel? How about the bottom tenth? How about the three molecules of gas in the exact center of the vessel? The pressure in the vessel is always P, independently of how much of the gas you are measuring."
5.) Linear mass density = mass/ length , can go up by increasing density, mass, or thickness of strings
6.) Like this point Princeton made about ideal gas intersecting at -273.5 Celcius and 0 Kelvins on P vs. T (or same could be said for V vs. T) graphs. The smaller slope line represents the compound with a greater relative molar mass. PV =nRT can be used to determine slope.

7. AAMC6 had question about terminology transitional vs. rotational vs. just "equilibrium" that made me understand it better.
Transitional equilibrium= sum of external forces is zero. Rotational equilibrium = sum of external torque is zero. And equilibrium is when a body is BOTH in transitional and rotational equilibrium.
8. Oh yeah, platelets (& mature RBCs of course) don't have nuclei and the origin of the coelom is the mesoderm (know it!) & The appendix is in the large intestine
9. Yes, like dissolves like except for a major exception: Acids dissolve bases and Bases dissolve acids (thank LeChatelier). Don't get it twisted.
So which of the following substances is most likely to be more soluble in 1.0 M HCl than in 1.0M NaOH: HI or Pb(OH)2? Pb(OH)2.
10.) Insulation for an electrical current is ALWAYS needed. If an answer choice suggest that: to make less insulation necessary or to minimize insulation, it's useless answer choice.
11.) Just like the catalyst increase the rate of a reaction, an increase in current and/or voltage in an electrochemical cell will increase rate at which a metal at the cathode is produced (not the amount--only amount of starting material can influence amount).
12.) High-yield reaction Converting a ketone ---> imine using hyrdrazone participating in a 2-step addition, elimination rx

13.) Epigenetic development of a gene: modulates gene expression such that one gene can lead to expression of several proteins--so more variation at the RNA level.
14.) Watch out for classic tell-tale sign of a recessive disease: 2 unaffected parents produce an affected child.
15.) Need to remember milliEquivalents equation = mg x valence / (formula weight).
What does an equivalent value tell? It gives you the number of univalent counter ions needed to react with each molecule of substance. So HCl has 1 equivalent of a mole meaning that 1 mole H+ ion is needed to react with 1 mole of a Cl- ion.
14.) telltale of a Sn1 or E1 reaction: rx takes place in acidic conditions. Carbocation can only exists in acidic solutions.
15.) nitric oxide can is capable of diffusing across the plasma membrane & no ions can diffuse across membranes-b/c of the polarity of ions, they need channels
16.) CCK is released by small intestine (by duodenum and jejenum) and acts on the gall bladder to stimulate release of bile.
17.) Double displacement = metathesis (when all ions switch partners kind of like the Shania Twain situation)
18.) Intermolecular forces, strongest to weakest: hydrogen bonding, dipole-dipole, london dispersion forces
But beyond that, hydrogen bonding is well understand. But what about dipole-dipole forces? This occurs when 2 polar bonds from two different molecules attract one another. I got a question wrong that helped m understand this: Dipoles arise from an electronegative bond involved in a polar bond (such as oxygen or nitrogen) pulls electron density from another atom also involved in a polar bond. London dispersion forces are fleeting, make-shift interactions that only arise from electron density temporarily shifting to one side of the molecule.
19.) Increasing the temp will ALWAYS increase rate of rx. So increase in temp = increase in rate of formation of product.
20.) Fats are absorbed into lacteals in the small intestine. Unlike carbohydrate and amino acids, they bypass the hepatic portal system (the liver circulatory system) and head directly into the system circulation via the thoracic duct. And once they're in the bloodstream, fats are packaged into lipoproteins.
21.) Important to look at solvents when determining nature of reaction. For example, Dimethyl sulfoxide (DMSO) is a polar aprotic solvent--boom w/ primary alkyl halide it's an SN2- concerted mechanism w/ neutral intermediate and bimolecular rx.
22.) Pretty much every major carbohydrate is an aldose, except fructose. Fructose is a ketose, whereas glucose, galactose and mannose are all aldoses. The same sugars in different optical families are enantiomers (e.g. D-glucose & L-glucose), all the other same-carbon sugars are disatereomers.
23.) An IR spectrum results when chemical bonds b/w two atoms absorb infrared radiation as a result of interatomic bind vibrations. To give a line in an IR spectrum, remember that the molecule must exhibit a change in dipole moment during vibration. (So Cl2, for example, would not produce an IR spectrum).
24.) This is a momentum problem, I took some time to do with math--but the math is not necessary! So I'll review the problem here.
"An 80 kg ice skater, standing on an essentially frictionless frozen pond, propels himself forward by throwing a 5 kg mass in the opposite direction with a speed of 1m/s. The skater is then propelled forward at a speed of 1/16 m/s. The total momentum o the skater and mass when both are moving is: 0 kg m/s.
The principle of conservation of momentum states that when the net impulse of the external forces acting on a system is zero, the total momentum of the system remains constant. So, I could have considered the skater the mass a system. Before the skater throws the mass, neither has a velocity and so the momentum is zero. The skater was standing and holding the stationary mass as well.
25.) Not new, but I keep forgetting. Primary spermatocytes are diploid. Primarty spermatocytes undergo meiosis I to form haploid secondary spermatocytes.
26.) AAMC listed these frequencies that are important: C≡N = 2250 , C-O: 1000-1300 , C=O : 1700-1850, O-H: 3200-3500
27.) Amides are formed from amines via a nucleophilic acyl substitution.
28.) Important to keep in mind that antibodies are proteins and that T-cells, unlike B-cells, do not recognize free floating antigens
29.) The most effective way to demonstrate necessity is to test for the occurrence of a specific outcome in the absence of the agent whose necessity is being tested.
30.) Macrophages can activate T-helper cells when it presents is antigens to T-cells, it also produces cytokines.
Last edited:
