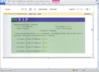
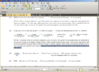
I did reply in mEq... look at the end of the answer...
How did you get 9 mOsm =27 meq?!
1mOsm= 0.0546gm so
9 mOsm= 0.491gm...
and 1 meq= 0.164gm
so if you do the work out u'll get 3... 27 is impossible...
Show me ur work out,if you please, but not the one written in the answers of the 120 STD questions...
My friend, you asked for a FULL AND DETAILED workout, so here it is, please be patient because I will write with lots of details:
++++++++++++++++++++++++++
PROFESSOR WROTE:
MW of Na phosphate = 164
How do we get above?
Knowledge: Na phosphate = Sodium Phosphate = Na + Na + Na + P + O + O + O + O = Na3PO4
GIVEN: Na = 23, P = 31, O = 16
Combined, we get: 23 + 23 + 23 + 31 + 16 +16 +16 +16 = 164
This means:
1 MOLE of Sodium Phosphate = 164 grams of Sodium Phosphate
This also means:
1 miliMOLE of Sodium Phosphate = 164 miligrams of Sodium Phosphate
This
DOES NOT mean:
1 OS-MOLE of Sodium Phosphate = 164 grams of Sodium Phosphate
This
DOES NOT mean:
1 miliOS-MOLE of Sodium Phosphate = 164 miligrams of Sodium Phosphate
With that in mind, let's continue:
++++++++++++++++++++++++++
PROFESSOR WROTE:
So, 164 mg = 3 millimoles of Na per millimole Na phosphate
How do we get above?
Knowledge: Na phosphate = Sodium Phosphate = Na + Na + Na + P + O + O + O + O = Na3PO4
That means:
1 MOLE of
Na phosphate = 3 MOLES of
Na
We found out earlier: 1 MOLE of Na phosphate = 164 grams of Na phosphate
Combined, we get:
1 MOLE of Na phosphate = 3 MOLES of
Na = 164 grams of Sodium Phosphate
This also means:
1 mili-MOLE of Na phosphate = 3 mili-MOLES of
Na = 164 mili-grams of Sodium Phosphate
With that in mind, let's continue:
++++++++++++++++++++++++++
PROFESSOR WROTE:
9 mMoles of Na phosphate (x) 3 = 27 mMoles of sodium
How do we get above?
GIVEN: WE WILL ADD INTO BAG: 9 mMoles of Na phosphate.
We found out earlier: 1 mili-MOLE of Na phosphate = 3 mili-MOLES of Na
Combined, we setup CROSS MULTIPLY like this:
WE KNOW: 1 mili-MOLE of Na phosphate = 3 mili-MOLES of Na
WE WANT TO KNOW: 9 mili-MOLES of Na phosphate = X mili-MOLES of Na?
TOOL: CROSS MULTIPLY, we get:
[(9 mili-MOLES of Na phosphate) MULTIPLY (3 mili-MOLES of Na)] THEN DIVIDE OVER (1 mili-MOLE of Na phosphate) = 27 mili-MOLES of Na
So, we know now:
9 mili-MOLES of Na phosphate = 27 mili-MOLES of Na
With that in mind, let's continue:
++++++++++++++++++++++++++
PROFESSOR WROTE:
1 mMole of Na = 1 mEq of Na because Na is monovalent
How do we get above?
Here is my formula, I memorized this for exam (Valence means Charge means +):
If 1 charge, then, 1 Eq= 1 MOLE.
If 2 charge, then, 2 Eq= 1 MOLE.
If 3 charge, then, 3 Eq= 1 MOLE.
+++++++
Side note: Is that hard to remember? What is my trick?
Eq can be thought creatively as
Excellent
Queen.
MOLE can be thought creatively as
Muscular John
If Muscular John has only 1
Power Charge, Muscular John will have only 1 Excellent Queen.
If Muscular John has only 2
Power Charge, Muscular John will have only 2 Excellent Queens.
If Muscular John has only 3
Power Charge, Muscular John will have only 3 Excellent Queens.
Now, to remember even better, we can substitute that
P Charge as symbol, remember symbol of charge is a symbol like this +
If Muscular John has only 1 +, Muscular John will have only 1 Excellent Queen.
If Muscular John has only 2 +, Muscular John will have only 2 Excellent Queen.
If Muscular John has only 3 +, Muscular John will have only 3 Excellent Queen.
NOW USE YOUR CREATIVE MIND, PICTURE a JOHN, with 3 +++, and YOU WILL NEVER FORGET THIS HORRIBLE JOHN.
(I am not going to draw out the picture, you have to imagine the picture.)
(I was explained like this and given the picture of this in college and the picture stuck with me to solve this Eq problem.)
Let's continue......
that also means:
If 1 charge, then, 1 mili-Eq= 1 mili-MOLE.
If 2 charge, then, 2 mili-Eq= 1 mili-MOLE.
If 3 charge, then, 3 mili-Eq= 1 mili-MOLE.
Therefore, automatically, we HAVE TO KNOW THIS AUTOMATICALLY:
because Na is monovalent,
1 mMole of Na = 1 mEq of Na
With that in mind, let's continue:
++++++++++++++++++++++++++
PROFESSOR WROTE:
Thus, 27 mMoles = 27 mEq of sodium added with the Na phosphate
How do we get above?
We found earlier:
9 mili-MOLE of Na phosphate = 27 mili-MOLES of Na
We know in our head: If 1 charge, then, 1 mili-Eq= 1 mili-MOLE.
Combined, we set up CROSS MULTIPLY like this:
WE KNOW: 1 mili-Eq of Na = 1 mili-MOLE of Na.
WE WANT TO KNOW: X mili-Eq of Na = 27 mili-MOLES of Na?
TOOL: CROSS MULTIPLY, we get:
X mili-Eq of Na = [(1 mili-Eq of Na) MULTIPLY (27 mili-MOLES of Na)] THEN DIVIDE OVER (1 mili-MOLE of Na)
X mili-Eq of Na = 27 mili-Eq of Na
Answer: 27 mili-Eq of Na
Meaning:
Question was:
How many mEq’s of sodium will be added as a result of using sodium phosphate?
Answer is: When we use 9 mMol of Sodium Phosphate, we will add 27 mili-Eq of Na.
Answer is: When we use 9 mMol of Sodium Phosphate, we will add 27 mili-Eq of Sodium.
Conclusion: 3 posters here agreed: in Packet 120 math question from SDN, the answer of question 69 was correct at 27 mili-Eq of Sodium.
(Posters: 3booda, Shahis77,molyhelp)
Good luck. And please help others. Thank you very much in advance.
[email protected]