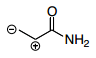
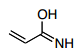
What are the options?
I'm not writing this down, so I may very well miss something, but here's how I see it:
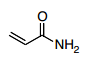
1st Resonance Structure
1.) The pi-bond on the oxygen forms a loan pair on the oxygen, making the oxygen negative and leaving behind a carbocation.
2.) The loan pair on the nitrogen forms a pi-bond with the carbocation causing the nitrogen to become positive and making the carbocation neutral.
3.) You have a positively charged nitrogen and a negatively charged oxygen with the same number of bonds as the orginal molecule. Going with what I said earlier in the thread, you have the same number of bonds but you now have charges on atoms which were previously uncharged species.
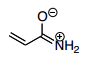
4.) Because the original structure has no charges and the same number of bonds as the resonance structure I described, the major contributor should be the one with less charges which is the original structure.
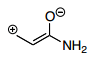
2nd Resonance Structure
5.) Another structure which could form would be to repeat step 1 from above then continue to step 6.
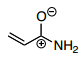
6.) Now, the pi-bond from the alkene moves over to form a pi-bond between the carbocation and the second carbon from the left. This leaves you with a carbocation on the far left and a negatively charged oxygen in the molecule which is again less favorable than the original structure meaning that it is also a minor contributor.
If I could draw it out in text I would.