I have a question about the portion underlined in red. I agree that 1M NaCl is much higher than 10 mM MgCl2. But it doesn't have Mg2+, even though it's as stable as those conditions that have Mg2+. So why is it so stable even though it lacks Mg2+?
I guess it's not because it has Cl- (not the right answer) - so why is it?
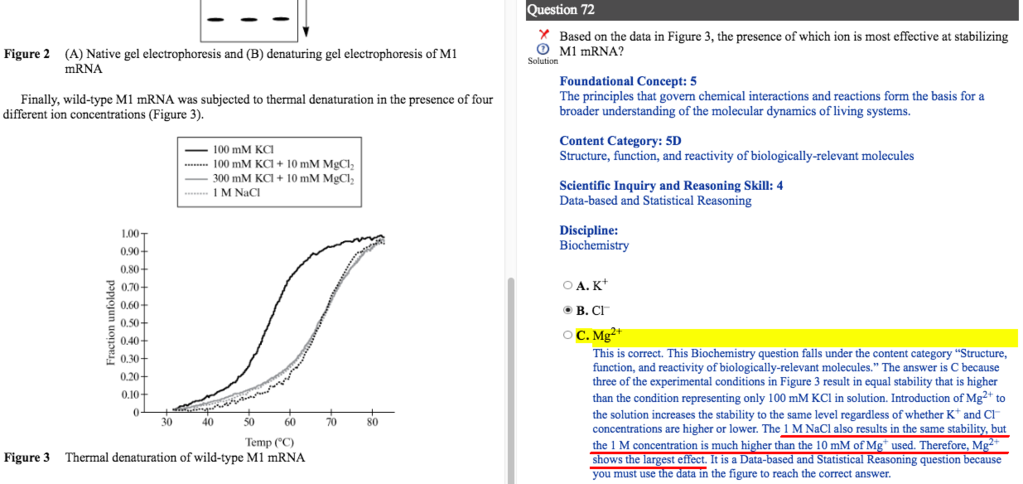
I guess it's not because it has Cl- (not the right answer) - so why is it?
