Advertisement - Members don't see this ad
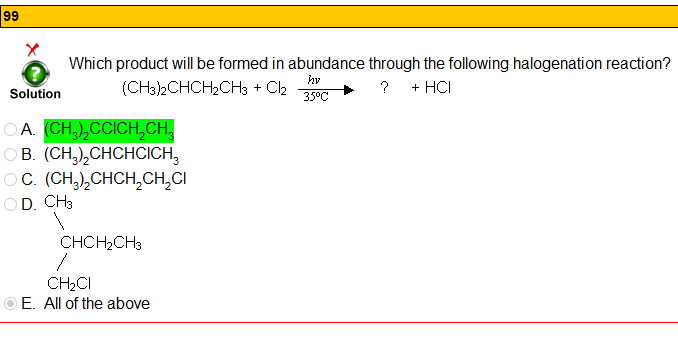
Soooo...I understand why their answer is correct but wouldn't theoretically all answers be correct?
Thinking about the Hammond postulate, isn't radical chlorination very non specific (unlike bromination) therefore yield primary, secondary and tertiary products?
I suppose my question is will the tertiary product be formed in abundance or will all products be formed in roughly equal amounts?
Thanks for the help!!
Thinking about the Hammond postulate, isn't radical chlorination very non specific (unlike bromination) therefore yield primary, secondary and tertiary products?
I suppose my question is will the tertiary product be formed in abundance or will all products be formed in roughly equal amounts?
Thanks for the help!!


