There was an APSF newsletter a good while back that described sugammadex, and referenced literature that it binds rocuronium at 3.57 mg/mg, meaning a 200mg vial of sugammadex reverses up to ~57mg of rocuronium. Unless you need reversal to occur very rapidly, you never need to overdose this ratio as far as I can see. I always ask myself, do I think there is more than 57 mg of rocuronium still floating around his patient? I have used the medication almost exclusively as my reversal agent for over 2 years without a single PACU respiratory complication, NMB related or not. Follow the link below for that ratio information.
New substances in our pharmaceutical armamentarium occur with reassuring frequency. When they tangibly affect our practice, they can be a tremendous

www.apsf.org
I respect that you're following advice from APSF but I STRONGLY disagree. The context of that ratio mentioned in the APSF article is a paragraph in a review article in Anesthesiology describing the molecule in theory, not the appropriate dosing. APSF recommended lowest possible dose due to concerns of supposed dose related likelihood of anaphylaxis. Appropriate dosing is later discussed in that same Anesthesiology article, at which point they recommend actual body weight dosing with the recommended doses we all know from the package insert. Additionally, they cite research saying even this can be inadequate, particularly when objective neuromonitoring is not performed.
-----------------------
This is the quote from the APSF article:
"With antibiotics, muscle relaxants, and latex, we expect and generally see reactions early in an OR case. Unlike these, sugammadex is typically administered at the end of a case. Thus a distinct difference is the timing of the anaphylaxis presentation and vigilance for anaphylaxis that may occur at what is historically an unexpected time for such an event. When sugammadex anaphylaxis happens, it seems to occur within 5 minutes of administration.5 Interestingly, the
likelihood of anaphylaxis with sugammadex appears to be dose-related.3 Therefore, it would make sense to use the lowest effective dose to decrease the incidence of anaphylaxis.
As an approximate rule of thumb, it requires 4 mg (3.57 mg to be exact) sugammadex to encapsulate/antagonize 1 mg rocuronium; thus a 200-mg reversal dose is adequate for most cases.6"
If you go source 6:
Residual neuromuscular block remains a significant patient safety concern. This article reviews the most common causes of residual block and offers suggestions for optimizing the use of muscle relaxants, facilitating their antagonism, and monitoring neuromuscular function.

anesthesiology.pubs.asahq.org
Sugammadex:
A decade ago, because of the limitations inherent in the use of acetylcholinesterase inhibitors as antagonists of neuromuscular block, it seemed clear that the issue of postoperative residual block was unlikely to disappear unless an alternative method of pharmacologic reversal of deep and even moderate block became available. In 2006, articles by de Boer
et al.80,
81 and others
82,
83 described a new and promising agent.
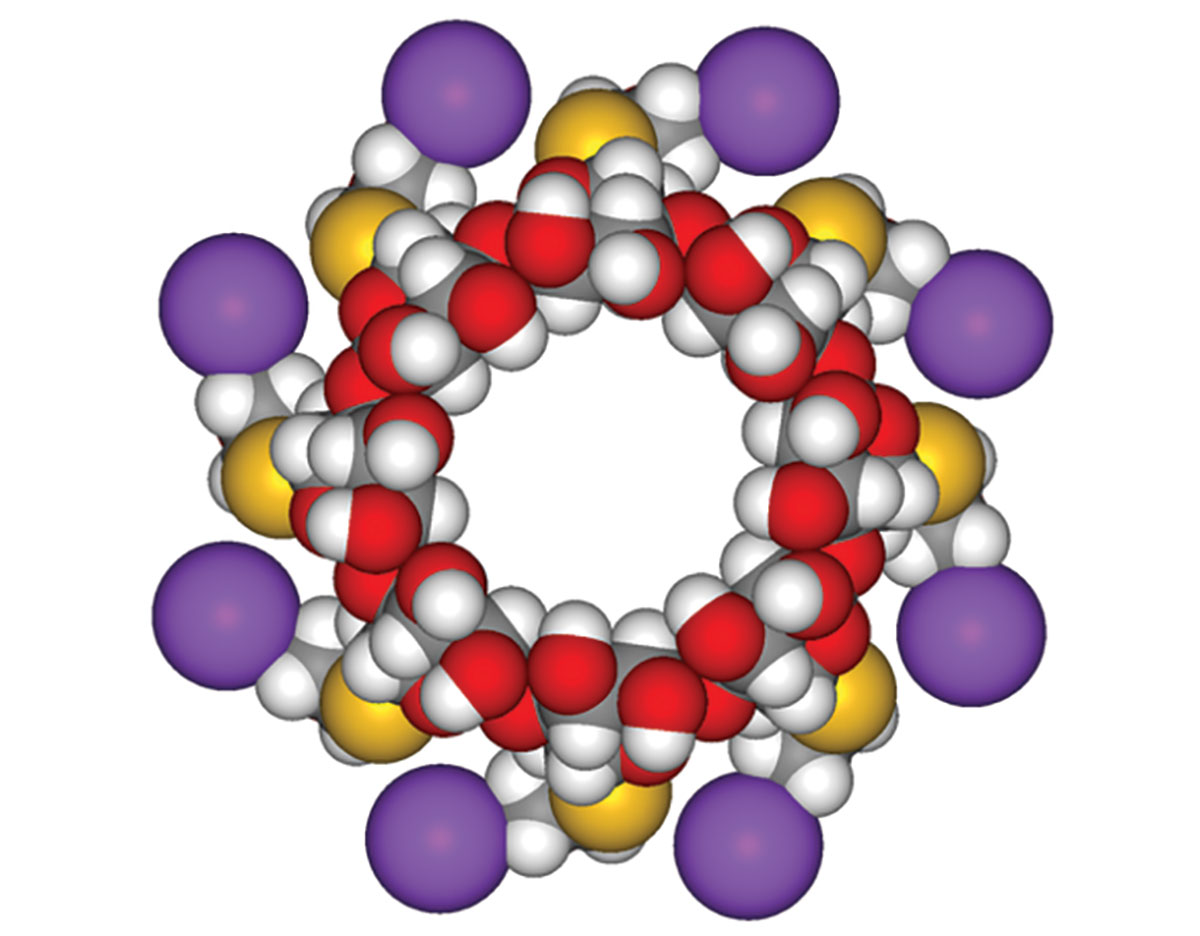
84 Sugammadex (a modified γ-cyclodextrin) forms 1:1 complexes with aminosteroid neuromuscular blocking drug molecules but has no effect on benzylisoquinolinium compounds or on succinylcholine.
A dose of 3.57 mg sugammadex is needed to encapsulate 1.0 mg rocuronium. The resulting complex has a very low dissociation rate. Encapsulated molecules of the NMBA that circulate in the plasma are no longer able to bind with muscle acetylcholine receptors, allowing blocking agents to diffuse away from the synaptic cleft and back into the plasma as the concentration of free drug in plasma decreases precipitously. Thus, clinicians now have for the first time the ability to rapidly and completely reverse profound nondepolarizing neuromuscular block, and do so directly by inactivating the activity of the NMBA, rather than indirectly, by inhibiting acetylcholinesterases.
Dosing:
These studies suggesting that residual neuromuscular block may still occur after sugammadex reversal need special mention. Because of the 1:1 molar ratio between sugammadex and the aminosteroid NMBA, there have to be sufficient sugammadex molecules administered to encapsulate all of the free molecules of the NMBA. For this reason, sugammadex reversal dose is calculated based on the depth of neuromuscular block at the time of reversal. For reversal of moderate block (TOFC, 1 to 3), a dose of 2 mg/kg is recommended; for reversal of deep block (PTC more than or equal to 1), a dose of 4 mg/kg is recommended, and for rescue from a failed rapid-sequence induction in the cannot-intubate-cannot-ventilate scenario (profound block, PTC = 0), a dose of 16 mg/kg is recommended (
table 3). Therefore, if clinicians use subjective or clinical means of assessment of neuromuscular block rather than objective monitoring, it is possible that the dose of sugammadex may be insufficient, resulting in incomplete reversal. This is likely a limitation of inappropriate monitoring rather than a failure of the drug.
20
It also appears that sugammadex should be administered based on actual body weight, particularly in the obese patient. Failure to administer a sufficient dose may result in reappearance of postoperative neuromuscular weakness (recurarization).101