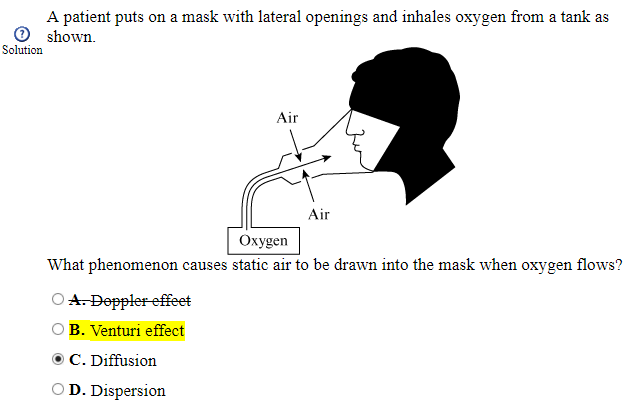
The AAMC is actually wrong here: Venturi masks actually work through the Bernoulli principle and constant pressure jet-mixing.
"Venturi masks entrain air by the Bernoulli principle and constant pressure-jet mixing.10 This physical phenomenon is based on a rapid velocity of gas (e.g., O2) moving through a restricted orifice. This action produces viscous shearing forces that create a decreased pressure gradient (subatmospheric) downstream relative to the surrounding gases. The pressure gradient causes room air to be entrained until the pressures are equalized."
Air moving down a " decreased pressure gradient downstream relative to the surrounding gases" is best described as DIFFUSION.
Source: Benumof and Hagberg's Airway Management (Third Edition)
You are right Vanturi masks do work based on Bernoulli principle but the Venturi Effect explains HOW this principle operates. I do not recall exactly what the Vanturi principle states word for word but to sum it up its basically saying that an increase in velocity due to decrease in the cross sectional area will also result in the decrease of pressure. Look at the mask, the cross section of the hose attached to the oxygen reserve is small, velocity of that oxygen is HIGH but pressure is significantly lower. Using that in conjecture with how gasses follow pressure gradients (via diffusion) basically what the Venturi mask does is introduce a new gas into the system (the mask) through a small hole open to the environment with a pressure that is higher than the pressure created inside the mask. Because the pressure of the air/oxygen outside the mask is higher than that of the dynamic (flow) pressure inside the mask, air mixes with the oxygen inside the mask via the process of entertainment. (yes that is what it's called ♀️).
While Bernoulli's Principle P(i) + 1/2pv(i)^2 = P(f) + 1/2pv(f)^2 *** is applied. And this is the most confusing about the solution they gave: Pair=Pstatic + 1/2rv^2. Anyone looking at that would be confused, like what the f happened to the other 1/2v^2 on the other side? Well that is because they assume that we know that this mask works similarly to a manometer or that we understand stagnation pressure/total pressure of a flowing fluid. Idk about you guys but I do not remember any of this from physics.
*** (yes this is correct, there is no "pgh" because there is no height difference)
Here is more on that dumb ass equation if you care to read about it:
Another way to conceptualize that equation is like this:
Stagnation Pressure = Pstatic + Pdynamic
Or even
Ptotal = Pstatic + Pdynamic
Stagnation pressure/ total pressure is the pressure of a fluid that would be exerted "if" the flow was brought to a stop without loss of mechanical energy. Or rather the point where the "dead spot" occurs, that is an obstacle is in the way causing flow to divert around the object w/o disturbing the flow itself. It is a localized, reduction of flow. There would still be pressure at that spot but not due as much to the velocity of the fluid. In a way this is also "static" when it comes to a stop but the difference is that this is also the TOTAL not just the pressure of the static gas. (this is the most confusing part of this question for me)
Static pressure of a gas is the pressure that a fluid has when it is either stationary or is measured while moving with the flow or "among" the steady flow. This is typically the gauge pressure. It is the pressure that is experienced into "every direction".
Dynamic Pressure of a gas is the pressure of the gas where the gas is moving, that is it is the representation of the kinetic energy of a fluid while it is flowing. WHY the 1/2pv^2 is used as it's notation. This is the difference between the "total" pressure of a fluid and the completely static version of it.
ANYWAY back on topic. The Venturi mask effect works and explains the modified Bernoulli's principal equation question because the total pressure of air (or stagnant pressure or even total pressure of oxygen) was greater than the pressure of the static, steady flowing oxygen in the mask.
Idk if you can tell but I really obsessed over this one, because pressures are just the mf worst.