You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
ICDs
Started by caligas
D
deleted171991
Yeah, it's always the same answer from the rep, but I suppose some day I could run across a an ICD that is programmed differently.
Last edited:
I had a case a couple months ago with a patient with an ICD for CABG. Couldn't figure out the manufacturer and patient didn't know. Called both STJ and Medtronic to no avail. Decided to put a magnet on it. ICD discharged several times while taking down the LIMA. Metronic rep informed us that his Cardiologist usually puts in Biotronik devices, so we called them and they deactivated the device before going on pump. I always make an effort to figure out who makes the ICD and have them come in. I would rather not depend on the magnet for many reasons, including the one above
Not to mention recommendations are to interrogate after placing a magnet on any CIED to insure pre-magnet settings are restored.
http://www.asahq.org/~/media/sites/...ment-of-patients-with-cardiac-rhythm-mgmt.pdf
http://www.asahq.org/~/media/sites/...ment-of-patients-with-cardiac-rhythm-mgmt.pdf
I spoke with some EP attendings about this issue and they recommended having every device interrogated. They noted that some small percentage of people will have the magnet function intentionally deactivated (for people that work in magnet factories, Dick Cheney, etc), and that that's reason enough to interrogate. That, and then they're those possibly unpredictable European devices like the aforementioned Biotronik.
That is some crazy paper.
I would much rather learn to use the 4 common programmers than attempt to memorize all can shapes and battery configurations.
What I've never understood is why don't all manufacturers make their implantables have at least their manufacturer brand labelled on or inside the device in such a way that it is radiographically visible? Is that really that hard?
Also, they should all have an audible tone when magnet response is achieved. But what do I know.
Also, they should all have an audible tone when magnet response is achieved. But what do I know.
What I've never understood is why don't all manufacturers make their implantables have at least their manufacturer brand labelled on or inside the device in such a way that it is radiographically visible? Is that really that hard?
Also, they should all have an audible tone when magnet response is achieved. But what do I know.
I recently found out how heavy those interrogation machines are. Jesus Christ
http://forums.studentdoctor.net/threads/pacemakers-aicds.591750/page-2
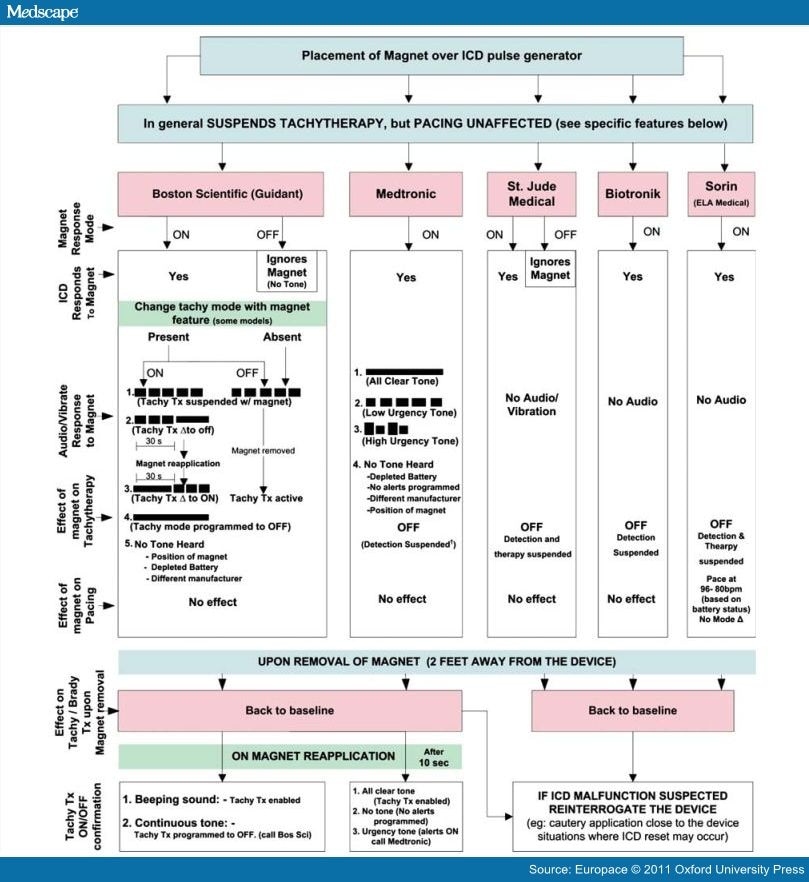
Medtronic and St Jude have about 90% of the market. My local reps all claim (in writing) that their devices are sensitive to a magnet turning off the AICD portion of the AICD/Pacemaker.
Medtronic and St Jude have about 90% of the market. My local reps all claim (in writing) that their devices are sensitive to a magnet turning off the AICD portion of the AICD/Pacemaker.
From MEDTRONIC- OFFICIAL WRITTEN DOCUMENT
Option 1: Medtronic's general recommendation for suspending therapies in Medtronic ICDs is to apply a Magnet or Smart Magnet ("Magnet") supplied by Medtronic. (See Magnet Use forSuspending Medtronic ICD Detection Standard Letter issued by CRDM Technical Services
Medtronic
U.S.) The Magnet, when placed over the device, suspends detection of rapid (tachycardia) electrical signals without affecting pacing therapy, eliminating the need for programming the device off. As soon as the Magnet is removed from the device, detection and therapies will return to previously programmed (non-Magnet mode operation) settings. No further programming with the removal of the Magnet is necessary. The presence of a Medtronic representative is not required for Magnet use. Medtronic is available to provide scheduled training and instructions prior to the first use of the Magnet.
a.The MCP is responsible for ensuring that the patient is constantly monitored for potential life-
threatening arrhythmias before, during and after the procedure.
b.The MCP is responsible for ensuring external defibrillation and pacing options are available to
deliver therapy should patient monitoring indicate a need for these therapies. If an arrhythmia is
detected, remove the magnet to return the ICD to normal detection and therapy delivery. The
Magnet response will result in temporary suspension of tachyarrhythrnia detection in all
Medtronic defibrillators
Option 1: Medtronic's general recommendation for suspending therapies in Medtronic ICDs is to apply a Magnet or Smart Magnet ("Magnet") supplied by Medtronic. (See Magnet Use forSuspending Medtronic ICD Detection Standard Letter issued by CRDM Technical Services
Medtronic
U.S.) The Magnet, when placed over the device, suspends detection of rapid (tachycardia) electrical signals without affecting pacing therapy, eliminating the need for programming the device off. As soon as the Magnet is removed from the device, detection and therapies will return to previously programmed (non-Magnet mode operation) settings. No further programming with the removal of the Magnet is necessary. The presence of a Medtronic representative is not required for Magnet use. Medtronic is available to provide scheduled training and instructions prior to the first use of the Magnet.
a.The MCP is responsible for ensuring that the patient is constantly monitored for potential life-
threatening arrhythmias before, during and after the procedure.
b.The MCP is responsible for ensuring external defibrillation and pacing options are available to
deliver therapy should patient monitoring indicate a need for these therapies. If an arrhythmia is
detected, remove the magnet to return the ICD to normal detection and therapy delivery. The
Magnet response will result in temporary suspension of tachyarrhythrnia detection in all
Medtronic defibrillators
Sure, but there are still recommendations to interrogate post magnet at the least.
Ideally you'd have the rep interrogate pre-procedure, deactivate tachy therapy and set to whatever asynchronous rate you want if pacer dependent and likely to be interfered with by EMI, and then return to prior settings postop.
The practice of simply using a magnet on every CIED because manufacturer spec sheets tell you the expected action and that there will be flawless return to baseline with magnet removal is potentially unsafe. It is possible for the magnet response to have been deactivated, the device to not respond as you expect, or failure to revert to pre-procedure settings with magnet removal (magnet/EMI can damage or reset existing settings, the response to magnet and/or magnet removal can be affected by battery level, etc).
It's simply a patient safety issue. Not to mention how it would look if you magnet deactivated for a surgery then removed the magnet and discharged the patient without interrogating the device and the patient had a bad outcome at home due to a change to or outright failure of device intervention/therapy.
Ideally you'd have the rep interrogate pre-procedure, deactivate tachy therapy and set to whatever asynchronous rate you want if pacer dependent and likely to be interfered with by EMI, and then return to prior settings postop.
The practice of simply using a magnet on every CIED because manufacturer spec sheets tell you the expected action and that there will be flawless return to baseline with magnet removal is potentially unsafe. It is possible for the magnet response to have been deactivated, the device to not respond as you expect, or failure to revert to pre-procedure settings with magnet removal (magnet/EMI can damage or reset existing settings, the response to magnet and/or magnet removal can be affected by battery level, etc).
It's simply a patient safety issue. Not to mention how it would look if you magnet deactivated for a surgery then removed the magnet and discharged the patient without interrogating the device and the patient had a bad outcome at home due to a change to or outright failure of device intervention/therapy.