- Joined
- Apr 29, 2011
- Messages
- 2,171
- Reaction score
- 863
- Points
- 5,246

When a sugar is provided in ring form (as a pyranose or furanose), how do you tell if it is L or D?
I know L or D comes from the last chiral carbon. This is easy to determine in Fischer projection. But what about when it's in a ring?

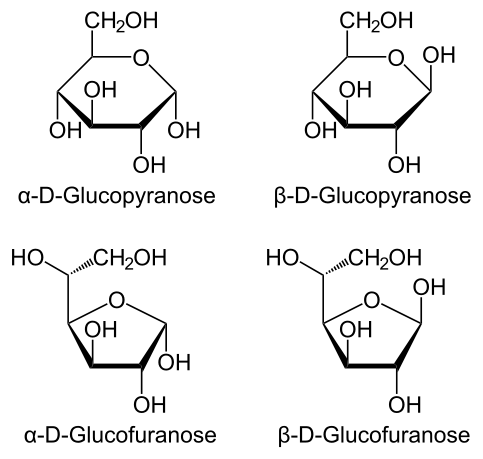
It's still no different. Remember, right or left is with respect to the -CH2OH group to the anomer itself, not your right or left. If it's right of the anomer, it's D. If it's left of the anomer, it's L. In all four scenarios above, -CH2OH is right of the anomer. Like I said, this is confusing because typically right/left is oriented with respect to the reader's right or left side. In this case, it's respect to the actual orientation on paper/screen. Unfortunately, that's the way it was taught to me. Maybe other's know an easier way, or you could just recognize that typically the -CH2OH group for D glucose points up and ignore everything else.@Czarcasm
With regard to
"If the -CH2OH substituent is RIGHT of the anomeric carbon, it's D.
If the -CH2OH substituent is LEFT of the anomeric carbon, it's L."
How does that apply here?
I think the anomeric carbon is the one on the right-most position (for each of those structures) and the CH2OH would be left of it. But these are all D.
The depiction I'm using is confusing, but that's how it was taught to me. Something that's probably more intuitive instead, is rather than focusing on where the -CH2OH group is appearing within the molecule, instead just look at the anomer itself:@Czarcasm

Yes, that's also something I learned in review books, but if you look at my earlier example, some people don't follow that convention. I'm not sure if it's because it's newer or what, but I suppose knowing both might be useful. The problem is, using either convention would yield different configurations.Hey czarcasm, I have been checking other sources, and some places say that D and L depends on whether the -CH2OH is above or below the plane of the ring
http://science.marshall.edu/castella/chm204/projection.pdf
Yeah, it's honestly not worth the added stress. I've never encountered a single question in practice asking me to specifically determine whether a haworth projection was D or L (only understanding D or L for fischer is of importance).Yeah youre right. I think I can push my neurotic mind away from this one. If there is more than a couple ways to do it, I doubt it'll be a problem. And even if it was a question on the MCAT, i bet it won't be a discrete. Thanks for everything Czarcasm
Are we supposed to know the position of the other -OH and -H in the ring structure for compounds like glucose, mannose or galactose?
I really wouldn't worry about this. It's a really low-yield concept.
I would beg to differ, based on my experience not too long ago. I can't talk about specifics, but suffice to say it was a very interesting passage on a useful case of an L-sugar.
The point being, there really is no such thing as high yield or low yield anymore.
I don't mean to make this an argument of semantics, but high frequency is not the same thing as high yield. Every exam is different and there will be only one (maybe two) questions on any highly specific topic. There may be two pH questions or two gas exchange questions, but each exam is designed to spread the questions out. So using your perspective for a given exam, all questions are technically low frequency on that exam. If we had a bigger sample size than two MCATs (one being old) for me and however many you took, we could reach a better conclusion about high versus low frequency of this particular material. But given that you and I have not taken every MCAT, neither of us can argue whether a topic is high or low frequency. We can speculate, but never say with confidence.
i've never saw someone show L and D like this(and my major is food engineering!) i'm not saying this is wrong (actually i am 🙂 )but there is a problem. we know that after the formation of the ring(only in Aldopyranoses), the O of H-C=O group is attached to 5th carbon and what is attached to other side of that O? the 1st Carbon. so in any Chemical structure for Glucopyranose we should have an Oxygen between 1st and 5th Carbon and that is not the case in this chemical structure. thats why i think it is wrong.
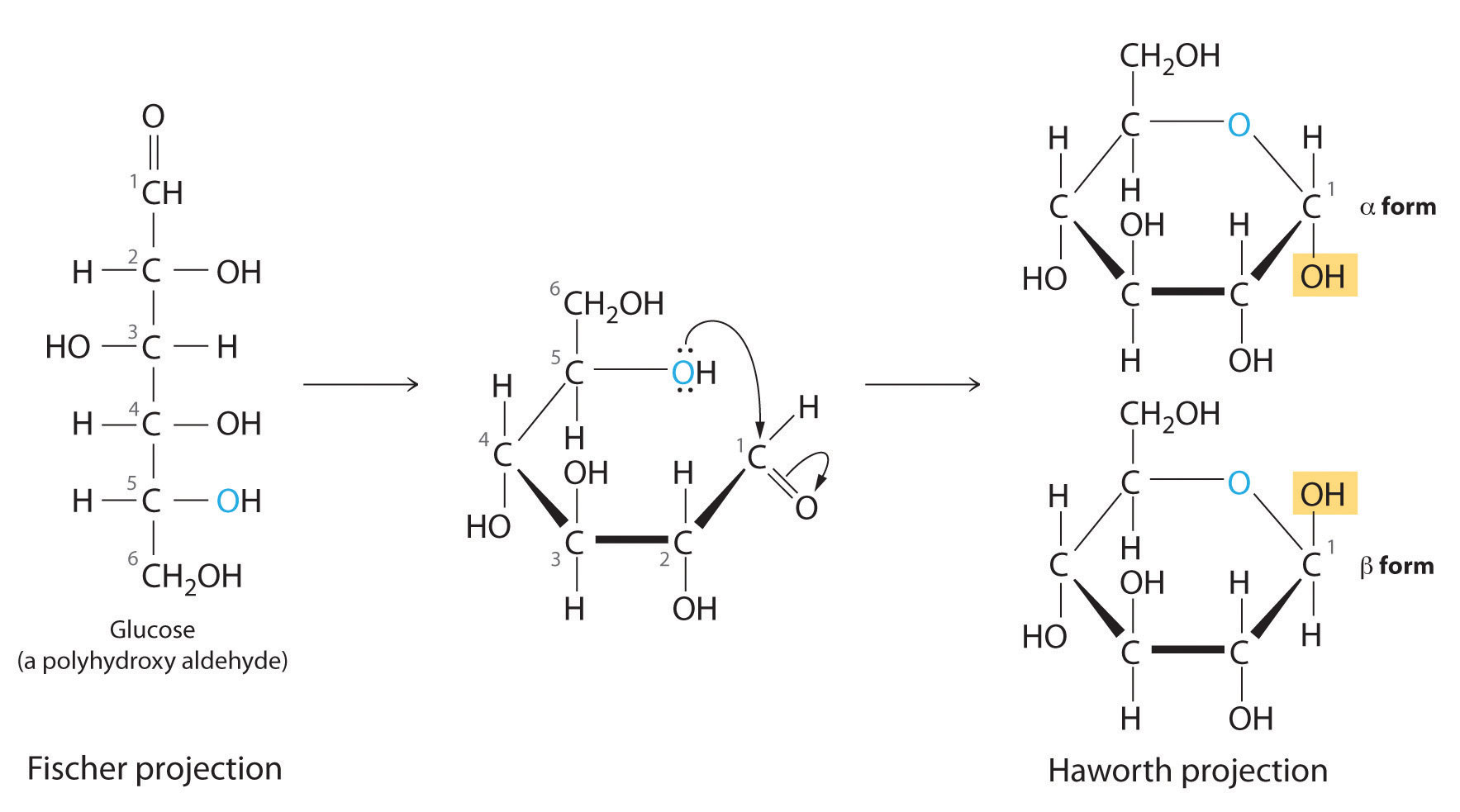
For the Haworth projection:
Hope this helps.
