- Joined
- Mar 7, 2009
- Messages
- 653
- Reaction score
- 0
- Points
- 0
- Dental Student
Advertisement - Members don't see this ad
I have been thinking about doing this for a while now but would like to compile all the periodic trends. I hear some people get ones with metals or something else that they have not seen before. Lets make sure we get them all!
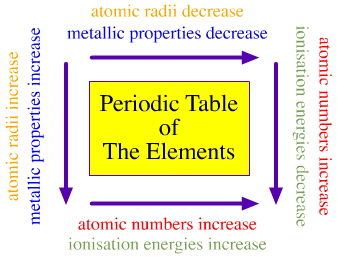
Question: Can you tell wich has the greater ionization? K+ or Ar?
Are they the same or does one have preference over the other?
Post more trends!

Question: Can you tell wich has the greater ionization? K+ or Ar?
Are they the same or does one have preference over the other?
Post more trends!
