MCATExamkrackers
New Member
- Joined
- Jan 10, 2019
- Messages
- 7
- Reaction score
- 1
Hi, I’m Austin, one of the MCAT instructors at Examkrackers. Our students in class often ask about phase diagrams and what makes water unique, so I wanted to provide an explanation that ties in with the recent polar vortex.
If you’ve looked at the news over the past week or so, you know that a polar vortex swept across the northern United States. Temperatures in Chicago, IL were reported to be -50ºF or below, and pictures of Lake Michigan showed ice floating on top.
MCAT Think: Why, as opposed to nearly all other liquids, does the solid form of water float? Take a look at the phase diagram for water below to give you a hint.
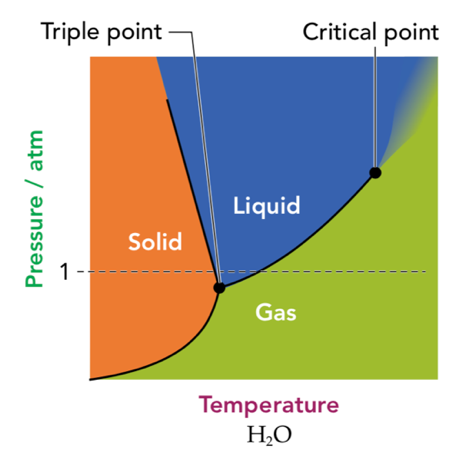
The slope of the line dividing the solid and liquid phases gives you a hint. As opposed to nearly all other liquids, the line has a negative slope. This means that, at a given temperature, a solid forms at a lower pressure than other substances. But what’s the molecular mechanism behind a floating solid?
A water molecule is made from an oxygen atom and two hydrogen atoms. Water molecules are attracted to one another by hydrogen bonds between the partially positive hydrogen and the partially negative oxygen. As water cools to below freezing, the hydrogen bonds re-orient to keep the partially negatively charged oxygens further apart (thereby minimizing electrostatic repulsion). Because density is mass per unit volume, the mass does not change, but the volume increases because of the spreading of the water molecules. This means that the overall density decreases, making ice float on top of denser water. Because the polar vortex significantly cooled the surface of Lake Michigan, this same phenomenon became visible to millions of Americans.
Hopefully this helps explain one of the chemical oddities induced by the polar vortex in the northern United States and gives you a way to remember why ice floats.
If you’ve looked at the news over the past week or so, you know that a polar vortex swept across the northern United States. Temperatures in Chicago, IL were reported to be -50ºF or below, and pictures of Lake Michigan showed ice floating on top.
MCAT Think: Why, as opposed to nearly all other liquids, does the solid form of water float? Take a look at the phase diagram for water below to give you a hint.
The slope of the line dividing the solid and liquid phases gives you a hint. As opposed to nearly all other liquids, the line has a negative slope. This means that, at a given temperature, a solid forms at a lower pressure than other substances. But what’s the molecular mechanism behind a floating solid?
A water molecule is made from an oxygen atom and two hydrogen atoms. Water molecules are attracted to one another by hydrogen bonds between the partially positive hydrogen and the partially negative oxygen. As water cools to below freezing, the hydrogen bonds re-orient to keep the partially negatively charged oxygens further apart (thereby minimizing electrostatic repulsion). Because density is mass per unit volume, the mass does not change, but the volume increases because of the spreading of the water molecules. This means that the overall density decreases, making ice float on top of denser water. Because the polar vortex significantly cooled the surface of Lake Michigan, this same phenomenon became visible to millions of Americans.
Hopefully this helps explain one of the chemical oddities induced by the polar vortex in the northern United States and gives you a way to remember why ice floats.
