Advertisement - Members don't see this ad
Why isn't this molecule a meso compound? Can't you draw a plane of symmetry from SH to oh?
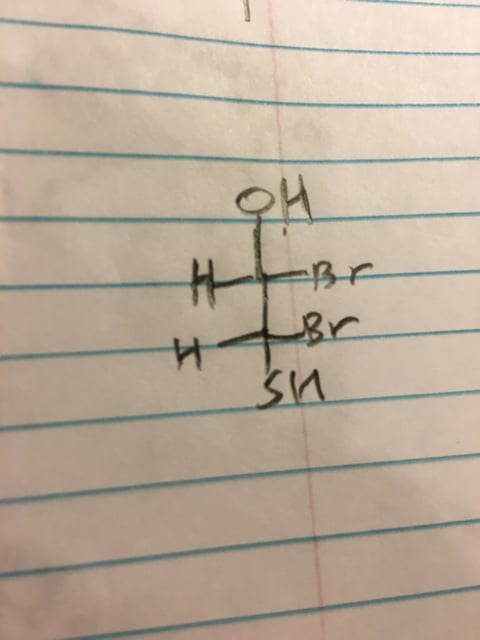
Sent from my iPhone using SDN mobile
Sent from my iPhone using SDN mobile

