Advertisement - Members don't see this ad
No question on #23, I'm just really ****ing pissed they made you round 50 down to 40. Are they serious? Is this the rounding that's done on the MCAT?! ****.

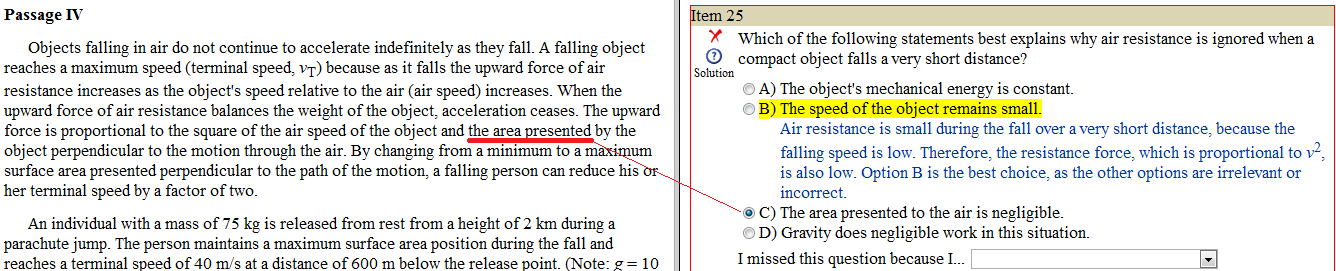
HUH?! I don't get these answers.
Also a question on #1:
Are oxidation-reduction products not effected by pressure?


HUH?! I don't get these answers.
Also a question on #1:
Are oxidation-reduction products not effected by pressure?

