Advertisement - Members don't see this ad
Just wondering if I'm thinking about this right? TBR didn't present it this way but I think this is a logical conclusion given that molar solubility is the maximum number of moles of solute that can dissolve in enough solvent to yield one liter of solution.
Question:
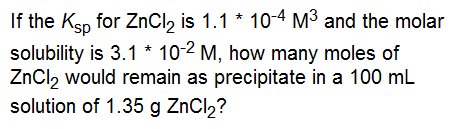
Answer:
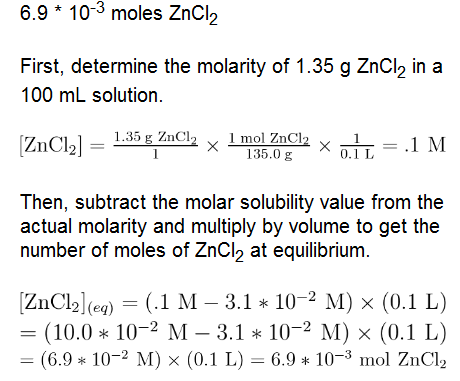
Question:
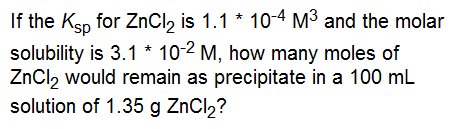
Answer: