You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
1H NMR spectrum
Started by nhasifuture
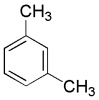
I'm gonna try my best to expalin this. Look at the attached picture of m-xylene and give each carbon on the ring a number. Start from the carbon on the top that has a methyl attached which would be carbon #1 then go clockwise...Anyways, both CH3's show a singlet and the hydrogen on carbon #2 shows a singlet also. Carbon #4 and #6 both show doublet and carbon #5 shows a triplet...Hope it makes sense!
Attachments
thanks, but i still dont see how carbon#6 shows a doublet and there is only 1 doublet on the answer. I thought C#6 would show a singlet.
oops, never mind I think i got it! Thanks again for a quick response.
Advertisement - Members don't see this ad
There is one Hydrogen on the carbon next to the 6 position. If its neighboring carbons have one H then n+1 is 2, two peaks or a doublet.
I'm gonna try my best to expalin this. Look at the attached picture of m-xylene and give each carbon on the ring a number. Start from the carbon on the top that has a methyl attached which would be carbon #1 then go clockwise...Anyways, both CH3's show a singlet and the hydrogen on carbon #2 shows a singlet also. Carbon #4 and #6 both show doublet and carbon #5 shows a triplet...Hope it makes sense!
What about Carbon # 3, I would expect it to be a sextet...or am I wrong.....??
Carbon #3 doesn't have any hydrogens!
oh...So N+1 applies only when there is at least a single Hydrogen present...not otherwise..!!
That clears it up for ......Thanks......!!
I would be really looking for carbon 1 and 3 in NMR, if you hadn't enlightened me...lol...Thanks again..!!
oh...So N+1 applies only when there is at least a single Hydrogen present...not otherwise..!!
That clears it up for ......Thanks......!!
I would be really looking for carbon 1 and 3 in NMR, if you hadn't enlightened me...lol...Thanks again..!!
Yeah make sure you look for Carbon 1 and 3 in 13C NMR but not 1H NMR 😀
Yeah make sure you look for Carbon 1 and 3 in 13C NMR but not 1H NMR 😀
Do we need to know 13C NMR for DATs....??
Advertisement - Members don't see this ad
Do we need to know 13C NMR for DATs....??
Indeed

To find peak character, use the formula n+1 where n=number of adjacent H's
RED H's: Split by 0 h's on adjacent carbon (0+1=1 Singlet)
GREEN H: Split by 0 h's on adjacent carbon (0+1=1 Singlet)
BLUE H's: Split by 1 adjacent grey H (1+1=2 doublet)
GREY H: Split by 2 adjacent blue H (2+1=3 triplet)
RED H's: Split by 0 h's on adjacent carbon -doublet
GREEN H: Split by 0 h's on adjacent carbon -sept ( 7)
BLUE H's: Split by 1 adjacent grey H (1+1=2 doublet)
GREY H: Split by 2 adjacent blue H (2+1=3 triplet
Which part i missed?
GREEN H: Split by 0 h's on adjacent carbon -sept ( 7)
BLUE H's: Split by 1 adjacent grey H (1+1=2 doublet)
GREY H: Split by 2 adjacent blue H (2+1=3 triplet
Which part i missed?
RED H's: Split by 0 h's on adjacent carbon -doublet
GREEN H: Split by 0 h's on adjacent carbon -sept ( 7)
BLUE H's: Split by 1 adjacent grey H (1+1=2 doublet)
GREY H: Split by 2 adjacent blue H (2+1=3 triplet
Which part i missed?
For the red one: there are no hydrogens on the adjacent carbon atoms so you only get a singlet!
For the green one: same thing as the red one! there are no hydrogens on number 1 and 3 carbons!
Which singlet peak would be more downfield, the red one or green?
The green by a drop i think.
Similar threads
- Replies
- 17
- Views
- 6K