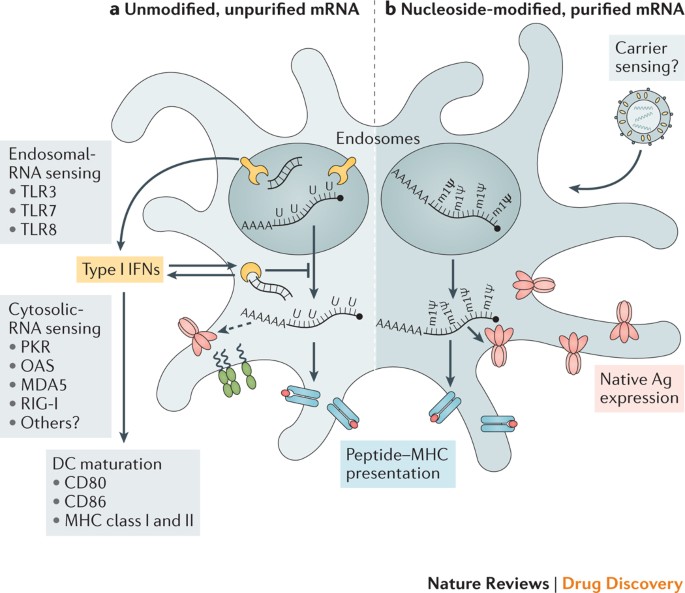
Several different mRNA vaccines have now been tested from phase I to IIb clinical studies and have been shown to be safe and reasonably well tolerated (
Tables 2,
3). However, recent human trials have demonstrated moderate and in rare cases severe injection site or systemic reactions for different mRNA platforms
22,
91. Potential safety concerns that are likely to be evaluated in future preclinical and clinical studies include local and systemic inflammation, the biodistribution and persistence of expressed immunogen, stimulation of auto-reactive antibodies and potential toxic effects of any non-native nucleotides and delivery system components. A possible concern could be that some mRNA-based vaccine platforms
54,
166induce potent type I interferon responses, which have been associated not only with inflammation but also potentially with autoimmunity
167,
168. Thus, identification of individuals at an increased risk of autoimmune reactions before mRNA vaccination may allow reasonable precautions to be taken. Another potential safety issue could derive from the presence of extracellular RNA during mRNA vaccination. Extracellular naked RNA has been shown to increase the permeability of tightly packed endothelial cells and may thus contribute to oedema
169. Another study showed that extracellular RNA promoted blood coagulation and pathological thrombus formation
170. Safety will therefore need continued evaluation as different mRNA modalities and delivery systems are utilized for the first time in humans and are tested in larger patient populations.
mRNA vaccines represent a promising alternative to conventional vaccine approaches, but their application has been hampered by instability and delivery issues. Here, Pardi and colleagues discuss recent advances in mRNA vaccine technology, assess mRNA vaccines currently in development for cancer...

www.nature.com