- Joined
- Nov 3, 2014
- Messages
- 2,872
- Reaction score
- 2,776
Created.
July 8 exam = August 9 release date
July 8 exam = August 9 release date
This is my third time writing the MCAT... I always seem to get 130-132 (or 13-15 on the old MCAT) in the sciences, but get 123-125 (5-8) on CARS...I honestly don't know what to do anymore, any suggestions?
Hey guys,
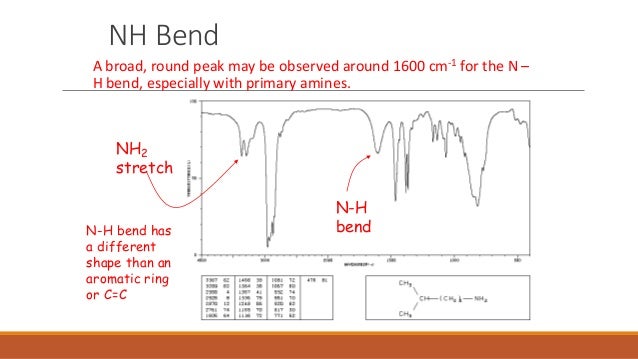
I'm stuck on this IR spectroscopy question from EK 2 for this molecule attached in the file:
http://www.chemsynthesis.com/molimg/1/big/1/1237.gif
The possible absorption wavenumbers are:
i. 3200-3500
ii. 1700-1750
iii. 1580-1610
I answered choices ii (carbonyl group) and iii (benzene ring), however, the correct choice was only choice ii (only accounted for the carbonyl group). Does anyone know why choice iii would not work?
Thanks!
That's what I thought too, but then I looked up the N-H stretch, and found out that N-H stretch is around 3200 too!
https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/spectrpy/Images/propacd2.gif
I'll try looking through the forums for EK tomorrow and try getting back to this. It's driving me crazy. Thank you for the response!
@Nucleophile1 The part in the passage that says "The Gsc transcript (generated from cDNA)" tells us that the transcript is mRNA, since it's generated from cDNA. In general, "primary transcript" usually refers to the transcription product, which is RNA.
Hi! Anyone have a concrete schedule for the July 22nd exam date? I would like to optimize my time since we only have ~7 weeks to go... and I'm getting nervous! During my spring semester I basically read through the books.. the past couple of weeks I've been using the Kaplan QBank as practice questions and this is definitely helping more than just reviewing content since that seemed to be too passive. If anyone has any schedule that they would share I would honestly appreciate it! Thank you!! Also, I already saw there are other schedules out there, but since we are all on the same timeline I'd prefer if any of you have something so I don't have to spend time modifying the other schedules. I seriously feel like I'm on SDN more than I should be....... since I should be studying right now... Thank you again!!!
How did you guys approach studying for the Psych/Soc section? What specific materials, and practice did you do?
Basically as a first time learner of the subjects when I read the material out TPR it felt like pure memorization.
Any tips on this would be appreciated. A bit worried about this section because I self studied it all.
I went through the Kaplan Psych/Sociology book and then did all of the psych/sociology questions on mprep.com. They have great questions for psych/sociology. It really helps solidifies all of the terms and theories.
Im using tpr stuff myself. A lot is remembering names and theories. Iv been reading a chapter and then doing passages accordingly and iv gotten better. Also the aamc flashcards are pretty good.Thanks. Never even heard of that resource lol. Did you do Khan problems? Are they comparable?
Thanks. Never even heard of that resource lol. Did you do Khan problems? Are they comparable?
Im using tpr stuff myself. A lot is remembering names and theories. Iv been reading a chapter and then doing passages accordingly and iv gotten better. Also the aamc flashcards are pretty good.
Yeah I think that site is a great resource specifically for sheer content knowledge. What I do is if I get a question wrong on that site, I put the topic into a Google Document to start compiling a list of topics I need to work on. I have also done some Khan Psych/sociology problems. The Khan problems are passages, whereas mprep seems to help more with content knowledge. If you are weak in content in that section, I would recommend pairing the mprep questions with the tpr book so that you can test yourself as you learn content.
Mostly tpr ones. Ill probably start doing some khan ones too.Thanks. Which passages though?
Got it. Sounds good. Thank you!
Is anyone taking the test July 8/9 or even July 22nd planning on applying to MD this cycle? I'm worried if it's too late…but really don't want to waste another year
I was doing the TBR Gases chapter and I came across this question. The answer is D but I think that B and D are both false. To say that gases are never in contact with one another is not true. The explanation even says that gases are in contact sometimes when they collide; they just don't stay in contact with one another. What do y'all think?
When they say gases are not in contact with each other, they mean they don't exhibit intermolecular forces. Gases behave independently of any other gas in the mixture and they exert their own partial pressure. By process of elimination, D would be the correct answer.
Is anyone taking the test July 8/9 or even July 22nd planning on applying to MD this cycle? I'm worried if it's too late…but really don't want to waste another year
@Nucleophile1 you have a good point- but assume the ideal case unless specified. The ideal conditions assume low pressure and high temperature. The ideal gas laws start to break down at high pressures and low temperatures.
@Nucleophile1 you have a good point- but assume the ideal case unless specified. The ideal conditions assume low pressure and high temperature. The ideal gas laws start to break down at high pressures and low temperatures.
Agree. Always assume ideal unless specifically specified not to. This is in the Guide. The word "never" is extreme, but clearly not as wrong as the other option.
Sent from my iPhone using SDN mobile
Can anyone explain number 47 on the sociology/psychology section bank? I don't understand the concept of intersectionality. Thanks in advance
Hi everyone,
Doing questions and reviewing the material now. Set to take the test July 22, 2016. When should I start doing the AAMC Q packs? What do you guys suggest? I have a full time job, but I'm leaving the last two weeks for full-time study. Should I leave the Q packs until then?
I am stuck on a Berkeley Review question. What do y'all think is the answer?
Well... we know low pKa = high acidity. Similarly, strong acid has a high Ka. It's saying that removing the base will result in shorter acting time. Since the passage refers to pKa and base concentration, my pick would be A: a higher pKa (more acidic) would result in lower [base]. Correct me if I'm wrong but it seems like every answer choice is saying the same thing? @aldol16
You're contradicting yourself here. A higher pKa is more basic, not more acidic. So here's what I think it's getting. At the anesthetic can be either protonated or deprotonated at the tertiary amine position. If it's protonated, it's cationic and that's what they're calling the "base." So whether it's cationic or neutral depends on its pKa. If it has a high pKa, it's going to be more basic even at lower pH. If it has a low pKa, it'll become neutral at higher pH.
So A is out because it's saying that the higher the pKa, the lower the base concentration when it should be the opposite. B looks good. C is out because high Ka means that the right side of the dissociation is favored, which means the conjugate acid is favored here (equilibrium in question is B-H ---> B- + H+). D is out because low Ka means the left side is favored, meaning the base (B-H) is present at high concentrations.
Just trying to reason this out. I may be entirely wrong.
Well... we know low pKa = high acidity. Similarly, strong acid has a high Ka. It's saying that removing the base will result in shorter acting time. Since the passage refers to pKa and base concentration, my pick would be A: a higher pKa (more acidic) would result in lower [base]. Correct me if I'm wrong but it seems like every answer choice is saying the same thing? @aldol16
You're contradicting yourself here. A higher pKa is more basic, not more acidic. So here's what I think it's getting. At the anesthetic can be either protonated or deprotonated at the tertiary amine position. If it's protonated, it's cationic and that's what they're calling the "base." So whether it's cationic or neutral depends on its pKa. If it has a high pKa, it's going to be more basic even at lower pH. If it has a low pKa, it'll become neutral at higher pH.
So A is out because it's saying that the higher the pKa, the lower the base concentration when it should be the opposite. B looks good. C is out because high Ka means that the right side of the dissociation is favored, which means the conjugate acid is favored here (equilibrium in question is B-H ---> B- + H+). D is out because low Ka means the left side is favored, meaning the base (B-H) is present at high concentrations.
I had the same thought process as @aldol16 but the answer is apparently A. They are saying the protonated cationic form is the acid.
Anyways, how ya'll feeling? I'm scheduled for July 8.
So far, in order of taking:
[5/21] NS1: 503 (126/124/125/128); 60% correct
[5/27] NS2: 507 (127/128/126/126); 66% correct
[6/1] AAMC unscored: 506-7 (estimated 126/126-7/126/128); 70% correct (64%/73%/63%/81%)
[6/8] NS3: 501 (wtf CARS?) (125/123/126/127); 55% correct
[6/14] NS4: 505 (127/125/125/128); 61% correct
Would like 1-2 more points in every subsection to be really comfortable. Definitely doable with 4 weeks left!
